
Which trends offer opportunities or pose threats on the European natural ingredients for health products market?
Europe's natural ingredients market for health products is changing fast. An ageing population and demand for preventive solutions drive these changes. Growth spans four segments: alternative medicine and natural remedies, preventive health products for immunity and digestion, functional beverages, and personalised nutrition. This creates opportunities for exporters from developing countries.
European buyers demand transparent supply chains with traceability, scientifically validated ingredients and AI-selected botanicals more and more. Companies who can meet stricter sustainability standards while delivering innovation will thrive. Bulk commodity suppliers, on the other hand, face transparency and pricing challenges.
Contents of this page
- Sector transformation
- Alternative medicine growth creates more demand for traditional botanicals and herbal remedies
- Preventive health drives growth in immunity, digestive and stress-relief botanical ingredients
- Stricter botanical regulations force suppliers to invest in quality systems and documentation
- Rising fraud concerns create opportunities for verified and traceable botanicals
- Format innovation, bioavailability enhancement and biotechnology reshape botanical ingredient requirements
- Functional beverages popularise botanicals and create market entry opportunities
- Personalised nutrition creates targeted demand for specific natural ingredients
- Sustainability certifications shift from competitive advantage to mandatory market entry requirement
- Increased need for traceability and transparency within supply chains
1. Sector transformation
The European natural ingredients market is becoming a ‘new market’ shaped by three forces. These are: demographic pressure from the ageing population, technological disruption caused by artificial intelligence, and increased sustainability and safety regulations. By 2035, suppliers will need to provide complete data packages, traceability documentation, clinical evidence, sustainability certification and standardised bioactive profiles for personalised nutrition formulations. The mass-market bulk commodity model is giving way to premium, data-verified, purpose-specific botanical ingredients.
The trends described show what changes the natural ingredients for health product sector will undergo in the coming years. In the next decade, the European market is expected to change in the following ways:
Higher demand for natural remedies
Due to its ageing population, European will have greater demand for health products that address chronic conditions. The appearance of personalised nutrition platforms means more consumers want alternative natural remedies that meet their specific needs. This drives growth in alternative natural remedies, leading to greater demand for natural ingredients. However, it is important to consider that, for pharmaceutical products, development, patents and data-driven formulation platforms will have higher standards in terms of efficacy and safety.
European consumers will be more aware of the negative side effects of synthetic pharmaceuticals products to their long-term health. This drives growth in alternative natural remedies and results in greater demand for natural ingredients. However, it is important to consider that pharmaceutical product development focuses on greater efficacy and fewer side effects, creating a threat to natural health products.
Stricter regulations for natural ingredients in health products
European companies will invest more heavily in traceability systems and requirements. They will require more detailled information at the production and manufacturing levels of producing countries. The increasing adoption of artificial intelligence in traceability systems is something to look out for. European companies face stricter regulations on sustainability obligations. It will also increase the need for data and other evidence on social and environmental impact in countries of origin. This results in the wider adoption of sustainability standards and requirements.
European authorities will have stricter systems and procedures to check the safety and efficacy of botanicals used in health products. Current developments show that botanicals are examined more strictly when it comes to their safety profile and possible impact on consumer health. Given the growing emphasis on consumer protection and consumer information, it is likely that there will be greater control on efficacy claims especially in relation to the food supplements industry.
2. Alternative medicine growth creates more demand for traditional botanicals and herbal remedies
European consumers are more and more concerned about the potential side effects of pharmaceutical drugs or regular medication. Many consumers are turning to alternative medicines, such as homeopathy, natural remedies and supplements. There is a growing awareness of the benefits of natural remedies in Europe. In 2024, the European herbal supplements market was valued at €7.8 billion. It is expected to grow to €15.9 billion by 2033. Europe made up 21% of the global herbal supplements market in 2024.
Healthcare professionals are promoting more alternative medicine, especially for patients with chronic health conditions. This has led to Europe becoming the largest global market for complementary and alternative medicine (CAM). The European CAM market was valued at €53.8 billion in 2024. It is expected to grow at an average annual rate of 21% between 2025 and 2030. Germany is the largest player in this market. It made up 20% of the revenue in 2024. This is due to its long tradition of natural health approaches, like homeopathy, herbal medicine and naturopathy.
The increasing interest in alternative medicine and natural remedies is an important driver for the natural ingredients market in health products. Consumers seek more natural approaches to improving their health and staying healthy. Companies respond to this by incorporating natural ingredients into their product range.
For example, Pukka Herbs (UK) uses natural ingredients like chamomile, ginger, turmeric and mint in their products. They teach consumers about the ingredients used in their products through the ‘Learn’ page on their website. They also sort their products by the qualities the ingredients have under the ‘Perfect for’ subsection of their ‘Shop & Learn’ section.
This rising demand influences major trends in the industry, such as the following:
- The shift toward increased examination of ingredients (see trend 4)
- A growing range of private-label products (see trend 7);
- A focus on sustainability (see trend 9);
- The search for new ingredients and natural health solutions.
Tips:
- Explore our study on the demand for natural ingredients in health products within the European market to gain more in-depth insights into major drivers of growth.
- Visit the website of Food Supplements Europe for more information on the European food supplementation market and specific consumer data. This website also provides an overview of national member associations and member companies in Europe.
- Visit the website of the European Herbal & Traditional Medicine Practitioners Association (EHTPA) to follow market developments, sector discussions and regulatory developments in Europe related to traditional medicine.
- Do your own research into the European market for health products to find opportunities for your specific botanical. Check online magazines such as Nutraceuticals World, Nutra Ingredients or Vitafoods Insights.
- For more information on research into the health benefits of your specific ingredient and to access scientific resources, visit Examine.
3. Preventive health drives growth in immunity, digestive and stress-relief botanical ingredients
The growth in the demand for different health products has created scope for botanical ingredients of interest to emerging market exporters. This is particularly the case within the food supplement sector.
Examples of interesting health product categories
Europe is a growing market for several health product categories. Some examples:
Immunity-boosting health products
The European market for immune-boosting health supplements was valued at €13.6 billion in 2024. It is expected to grow by 6.2% annually between 2024 and 2029. In 2023, France, Germany and Italy held about 30% of the European market share. These European countries have a strong natural health product industry and a growing demand for immune-boosting supplements. They play an important role in the imports of botanicals. The vitamin segment is set to see the most growth due to its antioxidant features that help fight infection. Products in this space include frankincense and turmeric in supplement form.
Digestive health products
The European market for digestive health products was estimated to be worth €13 billion and expected to grow at an annual rate of 7.3% from 2024 to 2030. Over 332 million people are thought to have digestive disorders in Europe, creating economic and social pressures. The highest rates are reported in Eastern Europe and less affluent parts of Western Europe. Germany, Italy, France, the UK, Spain and the Netherlands are the leading markets for natural ingredient exporters targeting digestive health.
Digestive health is an innovative market with many new product launches. For example, at Vitafoods 2024, German-based Symrise launched GutBalance+, a gummy food supplement that contained cranberry polyphenols. Supplements that contain turmeric, ginger, chamomile and fennel are often promoted as having digestive health benefits.
Cognitive health products
The European brain health supplements market was valued at €2 billion in 2023. It is expected to grow at an average annual rate of 12.5% between 2024 and 2030. Germany, UK, France, Spain and Italy are important markets. Memory enhancement, attention and focus, depression and mood, sleep and recovery, and stress and anxiety are notable sub-categories in this segment. Ginseng, ginkgo biloba, curcuma, gotu kola and lemon balm products are promoted for brain health.
Opportunities for lesser-known ingredients
Despite the opportunities, it is still difficult for exporters to hold a strong position in the European market. Especially when exporting ingredients that are less well-known than, for example, more established ingredients like fennel and peppermint (which are typically used in the digestive health segment). Achieving consumer recognition can be a long process.
An example of an ingredient that has secured its place in the market is baobab (Adansonia digitata). This fruit is harvested from the ancient Baobab tree native to Madagascar, mainland Africa and Australia. Baobab was approved by the Novel Food Regulation in 2008. Now it is commonly marketed within the European food supplement market. It is promoted as an ingredient that improves digestion, increases energy levels and supports the immune system and general health.
Despite overcoming a regulatory hurdle in Europe, baobab markets outside Africa remain underdeveloped. International consumers still need to be educated on its benefits and become familiar with its taste. For example, a 2018 survey of UK consumers showed that only 23% of consumers had heard of Baobab, of whom just 26% had actually tasted it.
Baobab is still relatively unknown in Europe amongst consumers in 2024. However, product launches with baobab have been rising slowly but steadily over the past decade. Brands such as Aduna (UK), Matahi (France) and Baola (Germany) continue to support and promote the product. They remain an important link between exporters and consumers.
Figure 1: Baobab products on the European market

Source: Terra Elements, Aduna and Health Essentials Direct, 2023
The age of Europe’s population puts pressure on healthcare systems. In January 2023, the EU’s old-age dependency ratio showed that 34% of the population was aged 65 or over. 28% of European consumers prioritised preventative healthcare, highlighting the shift towards maintaining quality of life in old age. Natural ingredients like frankincense are more and more important in multi-functional formulae for age-related conditions. Boswellia is used to support inflammation, joints and overall vitality. Products that combine Boswellia with turmeric, ginger and black cumin seed are marketed as ‘fighting the effects of ageing’.
Figure 2: Examples of products that contain Boswellia for joint strengthening

Source: Swanson, Sanct Bernhard, and Vegavero, 2025
Tips:
- See the CBI study on exporting immune-boosting botanicals to Europe for more information on this market and how to access it. Consult our studies on digestive health and stress & anxiety health products to learn more about those market segments.
- See the CBI study on exporting frankincense to Europe for more information on its applications in health products and how to access it.
- Promote your products and educate both consumers and buyers on their scientifically proven health benefits as well as their positive social and ecological benefits. Look at the website of the company B’Ayoba (Zimbabwe) for an example of simple and effective communication on both technical and impact data about their baobab products.
- Find out about the local practices in your country in relation to botanicals supporting specific health goals. Traditional uses can be a good starting point when choosing new ingredients. For new ingredients, check if there is a history of medicinal use of at least 30 years. 15 years of these years need to be in the EU. If you cannot find a history of safe use, find out if you can access the food supplement part of the market with your ingredient.
4. Stricter botanical regulations force suppliers to invest in quality systems and documentation
Botanical ingredients are constantly under investigation by European authorities due to their potential risk to consumers, restricting supply and product development opportunities. In mid-2024, experts from 26 national food safety agencies in the EU published a report on substances that should not be used or used to a limited extent in food supplements.
The 13 substances identified as posing a risk to human health include several commonly used in food supplements:
- St. John's wort
- Holy basil (tulsi)
- Cinnamon
- Turmeric
- Black pepper
This opens up the possibility for the EU to take steps to prohibit or restrict their use in the future, according to Article 8 of Regulation No. 1925/2006. Annex III in this article lists the current substances whose use in foods is prohibited, restricted or under investigation.
Beyond this list, the popular ingredient ashwagandha has been under investigation in Europe. There have been concerns about the safety and history of the use of ashwagandha in food supplements in the European market. According to a 2020 evaluation by the Danish Technical University, Ashwagandha can have a negative hormonal effect on reproduction in both animals and humans. Other studies have suggested additional side effects, including stomach upset, liver damage and very low blood sugar levels.
This has led to regulatory actions across Europe:
- Finland and Sweden are considering prohibiting the use of ashwagandha.
- France has warned against the use of ashwagandha by specific demographics, such as pregnant women and people under 18. This likely means that operators in France will need to start using new label warnings on food supplements that contain ashwagandha.
- The Netherlands has advised against the use of ashwagandha, among other ingredients.
- In Poland, maximum levels have been established for the daily intake of ashwagandha, stating a maximum 3 g of the root powder should be used per day and that the maximum with anolide content should not exceed 10 mg in the recommended daily portion.
- The UK has put ashwagandha on its Risk Analysis Tracker to identify and assess potential risks.
However, experts have disproved these statements and questioned the validity of the studies they were based on. One concern is about the parts of the plant which are used. Traditionally, the roots of ashwagandha are used. However, some manufacturers are using aerial parts of the plant in their product to boost yield and profitability. Doing this could introduce toxicity to ashwagandha products. The ashwagandha industry is actively defending the safety of standardised ashwagandha root extracts after Denmark’s risk assessment which may have failed to separate ashwagandha roots and leaves.
These cases demonstrate three important points:
- Food safety is an important issue in the European market. This means food safety assessments are constant. Exporters need to be prepared for careful examination.
- Technical dossiers are essential. They provide detailed information about your ingredient. This helps regulators evaluate its safety and suitability for use in food supplements.
- Complete documentation shows buyers that your product complies with EU requirements. It proves your product is safe for consumption and of consistent, high quality.
Have a look at the website of KSM-66 (India) to see how they ensure quality control for their buyers and how they communicate it.
The increased examination described in this section is not only a threat; it may provide opportunities and room for other botanicals with similar health benefits and established use in the European market. For example, some established ingredients in the digestive health category, like fennel, peppermint and ginger, could still obtain more market space
Tips:
- Stay informed on regulatory changes in the European Union. You can do so, for example, by following the food supplement page of the European Food Safety Agency and the food supplement page of the European Commission.
- Search the EU’s Rapid Alert System for Food and Feed (RASFF) database for examples of withdrawals from the market and the reasons for these withdrawals. These are related to ingredients used in the food and food supplements industry.
- Make sure you have a complete technical dossier on your product that you can present to potential buyers. O&3’s website(UK – natural oil sourcing) provides a good example of documents you should have. It includes detailed documentation for each of its product.
5. Rising fraud concerns create opportunities for verified and traceable botanicals
European buyers of natural ingredients for health products are constantly faced with lack of quality and safety control measures in the country of origin. Next to heavy metal contamination, adulteration (adding other things to ingredients, making them less pure) is a major issue that can have serious health risks for consumers of health products. Protecting consumers from misleading and potentially unsafe products has been a high priority on the European agenda for many years.
A study by the nonprofit ABC-AHP-NCNPR Botanical Adulterants Prevention Program (BAPP) that covered the North American, European and other international markets, reported on the adulteration rates for five botanicals used in health products. The table below show the ranking of adulteration among the five botanicals. It is important to note that products sold as licensed or registered herbal medicines have not been adulterated; the issue is focused on food supplements.
Table 1: Adulteration ranking of major botanicals used in health products
| Rank | Botanical ingredient | Scientific name | Common form | Approximate adulteration rate (%) |
|---|---|---|---|---|
| 1 | Ginkgo leaf | Ginkgo biloba | Extract | 56% |
| 2 | Black cohosh rhizome | Actaea racemosa | Root/Rhizome powder or extract | 42% |
| 3 | Echinacea herb/root | E. angustifolia, E. pallida, E. purpurea | Herb or root extract | 28% |
| 4 | Elderberry | Sambucus nigra | Berry extract or powder | 17% |
| 5 | Turmeric rhizome | Curcuma longa | Rhizome powder or extract | 16% |
Source: BAPP, 2024
Recent testing by EU authorities has confirmed that these concerns are ongoing. Between 2024 and 2025, a survey tested 104 commercially available cinnamon products from 13 European countries. It revealed that 66% of the samples did not meet international standards, were not compliant with European food safety legislation, were suspected of fraud or may have been toxic for children due to high coumarin content.
Common adulteration methods vary by ingredient:
- Curcuma (Curcuma longa): Illegally improving colour with azo-dyes or inorganic materials (yellow chalk, lead chromate). Also adding fillers (maize or rice flour);
- Cumin (Cuminum cyminum): Presence of mahaleb (related to almonds), peanut shells and almond husks;
- Cinnamon: Substitution of high-quality Ceylon cinnamon (Cinnamomum verum) with lower-quality cassia varieties (Cinnamomum aromaticum, C. burmanii, C. tamala). Also adding bark and other plant parts.
Adulteration is closely linked to sustainability and wild-stock depletion. This is because harvesting wild medicinal botanicals is becoming less sustainable. It puts pressure on supply and leads to more adulteration. Around 27 European medicinal plant species are endangered or near-threatened. For example, Boswellia serrata and Rhodiola rosea show both supply stress and increased adulteration risks.
There are growing authenticity and purity issues in the sector. In response, many laboratories around Europe, such as Eurofins Genomics, have increased testing to uncover fraud in botanicals production. Common methods include DNA analysis, isotopic techniques, mass spectrometry, spectroscopy, chemometrics and a combination of detection methods. Buyers are also more often demanding that exporters have (certified) quality and food safety management systems in place, next to improved traceability systems.
Tips:
- Refer to existing guidelines and other publications that can help you address the blind spots in your supply chain. For example, the Guidance on Authenticity of Herbs and Spices. This is published by BRCGS, the UK Food and Drink Federation and the Seasoning and Spice Association. It brings a collection of good practices to target adulteration issues. The guide describes diagnostic, preventive, detection and verification measures that suppliers can implement at different steps in the chain.
- Review digital technologies that can also offer you solutions to authenticity issues. Read our study on blockchain technology for agricultural ingredients to learn more about traceability systems and platforms that can help you map out all steps and actors in the chain, creating transparency and ensuring trust.
- Create trust-based and transparent relationships with buyers. This includes clear and fast communication, the fulfilment of promises and an effective response to non-compliance situations. Sending representative samples, which match the product variety and quality that you can supply, is also extremely important to prove that you are a reliable supplier. In CBI’s study on doing business in the European market for natural ingredients for health products, you can find some more tips that can help you build long-term relations with buyers.
- If you harvest wild botanicals, implement sustainable harvesting practices. This ensures long-term supply and reduces the risk of adulteration. Consider obtaining FairWild certification to show that you have a sustainable wild collection.
- Watch this video on food fraud in herbs and spices to get a better idea of how and why herbs and spices are adulterated.
6. Format innovation, bioavailability enhancement and biotechnology reshape botanical ingredient requirements
The EU invests heavily in sustainable food systems. This supports much of Europe’s innovation in natural health products. Through its Food 2030 initiative, the EU is driving research and development that aligns with the European Green Deal, Farm to Fork Strategy and Bioeconomy Strategy. The EU is investing €25 billion in 'Excellent Science' funding between 2021 and 2027, providing grants for frontier research, supporting researcher mobility and enabling collaborative projects.
Increasing investments in innovation and Research & Development (R&D) make Europe an interesting market for suppliers of natural ingredients for health products. The European health product industry is constantly investing in new product development, extraction technologies and research on medicinal plants. Countries such as Germany (Schaper & Brümmer and Dronania) and Sweden (New Nordic and Apoteum) are home to innovative health product companies. In the coming years, the ways to use natural ingredients in health products is expected to rise.
There are three big innovation trends emerging:
Format innovation combined with sustainability
The industry is making botanicals more accessible and convenient for consumers through new formats like ready-to-drink herbal teas, gummies, capsules and powders.
For example, Benicaros (the Netherlands) produces health products from upcycled carrot peel, which is rich in fibre. They offer products in gummies, powders and ready-to-drink beverages, combining convenience and circular economy principles.
Scientific validation and combination formulas
Most herbal products focus on specific health goals, such as reducing stress, improving digestion or enhancing sleep quality. These holistic health benefits resonate more with consumers than targeting isolated symptoms. Companies strengthen their products through scientific validation and combining ingredients.
For example, Pharmanager’s (France) Rhodiola rosea extract undergoes rigorous clinical testing to demonstrate effectiveness for stress management, and mental and cognitive performance. Companies like Monteloeder (Spain) combine hibiscus and lemon verbena extracts, scientifically proven to support metabolic health and weight management.
Biotechnology
Increasing investment in biotechnology and synthetic production methods may pose a threat to natural ingredient producers in emerging market economies. Spirulina Nord grows microalgae spirulina in controlled bioreactors with optimal light, temperature and nutrients, ensuring consistently high quality year-round.
As an example of what’s possible, Ayana Bio (US) uses plant cell technology to produce bio-actives found in traditional herbs like lemon balm and echinacea. Plant cells grow in controlled stainless steel tanks, minimising risks of soil contamination, pests and adulteration. However, these technologies remain very expensive, making it unlikely they will pose a great threat in the near term.
European innovation creates opportunities for informed exporters. You can use scientific validation and traditional evidence in your marketing. Understanding format trends, sustainability demands and scientific requirements also helps your ingredients to stand out and attract European buyers.
Figure 3: Example of stress relief gummies on the European market

Source: Bloom Robbins, 2024
Tips:
- Stay up to date on new innovation and developments in the European market, for example by accessing market information from sector associations such as the European Federation of Associations of Health Product Manufacturers. Another good source is Nutrition Insight, a news provider for health and nutrition, sustainability, and the global nutraceutical market.
- Make sure to provide complete documentation and specifications of your ingredient(s) to European buyers, including technical data sheets, and dossiers when requested. This information will allow them to assess the potential of your product in different applications.
- Read the Vitafoods Europe Trend Guide for insight into the latest product innovations and market trends in the natural health products sector. It is an annual report that outlines what European buyers and manufacturers are working on.
7. Functional beverages popularise botanicals and create market entry opportunities
European consumer behaviour is changing a lot. 71% of Europeans now drink less alcohol in 2025, wanting healthier alternatives for relaxation, socialisation and wellness. This creates large demand for functional beverages that offer natural solutions without alcohol or synthetic ingredients. Consumers are turning to traditional herbal teas, ready-to-drink botanical beverages and adaptogenic coffee alternatives. This creates good opportunities for natural ingredient exporters.
The European functional beverage market is expected to grow from €29.9 billion in 2025 to €41.5 billion by 2030, with an average annual growth rate of 6.78%. Energy drinks currently dominate the market, while sports hydration drinks are experiencing notable growth amongst fitness enthusiasts.
Three main segments offer opportunities for botanical ingredients:
- Traditional herbal teas: Hot infusions that contain botanical benefits. Available in familiar formats like tea bags and loose leaf.
- Ready-to-drink beverages: Convenient cold drinks, including botanical waters, functional shots and herbal iced teas.
- Adaptogenic coffee alternatives: Caffeine-free or low-caffeine beverages that combine familiar coffee formats with functional mushrooms and adaptogens.
Traditional herbal teas
European tea companies have been developing herbal and fruit tea blends that address specific health areas since the 1990s, creating demand for functional botanicals. Germany, Poland, the United Kingdom and France are the main European markets.
German-based Yogi Tea's Women's Energy combines hibiscus, liquorice, raspberry leaves and ginger for menstruation-related complaints. UK-based Pukka Herbs' Peace tea blends chamomile, spearmint, ashwagandha, liquorice root and lavender for relaxation and anxiety reduction. Both brands build on Ayurvedic principles in their formulations.
Supermarket brands have also tapped into this trend with private-label herbal infusions. French retailer Carrefour's Infusion Digestion Légère combines star anise, anise, fennel and liquorice for digestive support.
Note that the European Food Safety Authority (EFSA) needs to evaluate claims for botanical ingredients in teas and herbal infusions.
Figure 4: Example of a herbal tea for relaxation on the European market

Source: Clipper (UK), 2024
Ready-to-drink beverages
The market for ready-to-drink (RTD) botanical beverages is growing quickly. Consumers want convenience and pay premium prices for products that require no preparation. RTD formats include cold herbal teas, sparkling botanical waters, functional shots and botanical-infused energy drinks.
For example, UK-based PerfectTed produces matcha-based ready-to-drink beverages. These combine the energy benefits of green tea with natural ingredients like vanilla. The brand has successfully positioned itself in mainstream retailers. This shows how botanical beverages can compete with conventional soft drinks.
Another example is MOJU ginger shots, stocked by British supermarket Sainsbury’s. These shots are promoted as a “caffeine-free kick when your mind and body need it most”. They contain vitamin C, which helps reduce tiredness and fatigue and supports the immune system. These products show how supermarkets are supporting the shift towards natural, functional beverages.
Figure 5: Examples of ready-to-drink functional beverages on the European market

Source: Perfected and Moju, 2025
Adaptogenic coffee alternatives
The coffee alternatives and adaptogenic beverages segment is growing fast. These products combine familiar formats with functional botanicals to address stress, focus and energy issues without the side effects of caffeine. For example, London Nootropics’s adaptogenic coffee blends contain lion’s mane, rhodiola rosea, ashwagandha, cordyceps, Siberian ginseng and chaga mushrooms. These products target professionals who want improved cognitive performance through natural ingredients.
Figure 6: Examples of adaptogenic coffee on the European market

Source: London Nootropics, 2025
This trend creates a range of opportunities for suppliers of botanical ingredients. Functional beverages offer lower regulatory barriers than supplements, as many botanical teas and drinks do not require Novel Food authorisation. The variety of formats (hot teas, cold drinks, shots, coffee alternatives) means ingredients can reach consumers through multiple channels. As consumers become familiar with botanical benefits, they are more likely to purchase these ingredients as supplements. This creates demand in other categories.
Tips:
- Identify the chemical and nutritional profile of your natural ingredients, especially if they can be used in self-care and wellness products. Ensure you promote these features to buyers, for example by informing them and mentioning these properties in your marketing materials.
- Ensure you can substantiate your claims with scientific data and certifications. Do not make medicinal claims.
- Visit or participate in trade fairs to find out whether the market would be open to your product, get market information and find potential buyers. The most relevant trade fairs in Europe are SANA (exhibition of organic and natural products in Italy), Food Ingredients Europe (a leading international exhibition for food ingredients in Germany), BIOFACH (dedicated to organic products and hosted in Germany), Natexpo (international trade show for organic products in France) and Vitafoods (a nutraceutical trade show taking place in Spain in 2025, following its long-standing presence in Switzerland).
- Monitor the ‘sober curious’ movement and alcohol-free beverage trends. Botanical ingredients that can replace alcohol's complexity and ritual offer good opportunities.
8. Personalised nutrition creates targeted demand for specific natural ingredients
The European personalised nutrition market was valued at €2.55 billion in 2024. It is projected to reach €8.77 billion by 2033, growing 14% annually. This growth reflects a shift from one-size-fits-all supplements to data-driven, individualised nutrition solutions. Companies like Nourished use patented 3D printing technology to create personalised gummy vitamins. These use seven different nutrients, based on customer questionnaires about diet, lifestyle and health goals.
Vitl uses online health quizzes about what areas consumers want to improve like sleep, digestion, immunity and stress. It combines these with optional DNA and blood tests to create unique supplement formulas.
Established supplement companies are investing in this heavily. Nature's Way acquired Baze in 2020. Baze offers at-home blood tests that measure micronutrient levels. It then recommends custom supplements and food plans that have eliminated 73% of customers' nutrient deficiencies within three months. By 2033, the Nordic region is forecast to become Europe's fastest-expanding personalised nutrition market, reaching €477 million, while supplements lead all product categories.
This trend creates opportunities for natural ingredients exporters. Personalised nutrition companies need standardised botanical extracts with consistent potency, detailed certificates of analysis and full traceability. These requirements reward quality over volume. The integration of artificial intelligence (AI) helps get real-time feedback, driving demand for specific bioactive compounds proven to help with individual deficiencies. Adaptogens (like ashwagandha for stress), nootropics (like bacopa for cognitive function) and digestive botanicals (like ginger) are more often selected in precise combinations for individual needs.
In the short term (one to three years), expect personalised nutrition brands to look for smaller, more frequent ingredient orders as they scale custom formulations. Long term (beyond three years), demand will shift toward botanicals with documented effects on specific biomarkers. For example, blood sugar regulation, inflammation markers or microbiome composition.
Tips:
- Think about getting B-Corp or another sustainability certification. Personalised nutrition brands typically target health-conscious consumers who value ethical sourcing.
- Stay up to date on clinical research on your main ingredients with websites like PubMed, Google Scholar, ClinicalTrials.gov, NutraIngredients and Natural Products Insider because personalised nutrition companies rely on evidence-based formulations.
- Research which natural ingredients personalised nutrition brands from your region already use. For example, Nourished's ingredient range includes ashwagandha extract for energy and concentration, ginger extract for antioxidant support and nausea relief, and turmeric with black pepper. All of these are sourced from developing countries. Study the health benefits these brands promote to understand market positioning.
9. Sustainability certifications shift from competitive advantage to mandatory market entry requirement
The EU is committed to environmental sustainability and sustainable growth, something it has made clear in the European Green Deal. Sustainability is important in the European health products market, on several levels.
As a result, ethical sourcing has become a legal obligation. With the introduction of the Corporate Sustainability Due Diligence Directive (CSDDD) and the Corporate Sustainability Reporting Directive (CSRD), European companies now need to identify, prevent and address human rights and environmental risks in their supply chains. This means buyers require more documentation and transparency from their suppliers.
On an industry level, sustainability has become a central focus in the European food supplements industry. This is driven by both individual companies and through a coordinated, industry-wide strategy. The industry association Food Supplements Europe is leading the way. Its members have signed the EU Code of Conduct for Responsible Food Business and Marketing Practices.
European health product companies are becoming much more involved in sustainably managing the natural resources they use. European buyers may request that you meet the standards of their code of conduct or sign an industry charter. For example, see the Responsible Sourcing Policy of multinational dsm-firmenich to see what this entails for them and what they expect from their suppliers.
Beyond certification, platforms like Sedex are also popular for managing ethical sourcing. Sedex makes ethical sourcing practical by providing data tools and expert teams that help businesses understand risks, and act and show progress across supply chains.
On a consumer level, Europeans are also demanding sustainable products and are willing to pay premium prices. Studies show that 22% of Germans and 28% of Italians are prepared to pay more for sustainably produced food. One signal for sustainably produced food is organic, which are perceived as safer and of higher quality. This is driving demand for organic ingredients and creating opportunities for certified producers.
The global organic market was valued at €124 billion in 2023. The biggest opportunities for organic retail sales in Europe are shown in the figure below.
Source: FiBL, 2025
Per capita spending on organic ingredients is highest in Switzerland (€468 per person), followed by Denmark and Austria. In terms of market share, Denmark leads at 11.8%, Switzerland at 11.6% and Austria at 11%. Retail sales have increased by a lot in Estonia (13%) and the Netherlands (12.5%).
There are 4.3 million organic producers worldwide. India is home to the most, at 2.36 million. Among countries exporting natural ingredients for health products, Vietnam, Sri Lanka and Burkina Faso have shown particularly strong growth in organic producer numbers.
Brands like Pukka Herbs show the value of transparency in building consumer trust. Their website shows traceability throughout their supply chain, showcasing the farmers and communities they work with, displaying certifications like Soil Association, FairWild, Fair for Life, Halal, Kosher and Organic prominently and explaining their sourcing practices. This level of transparency is more and more expected by European consumers and can help your products stand out in the market.
To make good use of this opportunity, exporters should consider if they have a business case that can prove this and/or obtain extra certification. For example, you can show you practice sustainable sourcing by using Good Agricultural and Collection Practices (GACP) for medicinal plants. You could also consider obtaining certification such as FairWild. This certification is proof that you source your ingredients ethically and sustainably. Note that initial certification fees as well as renewal fees in the future can be costly.
Figure 8: Example of sustainable sourcing roadmap
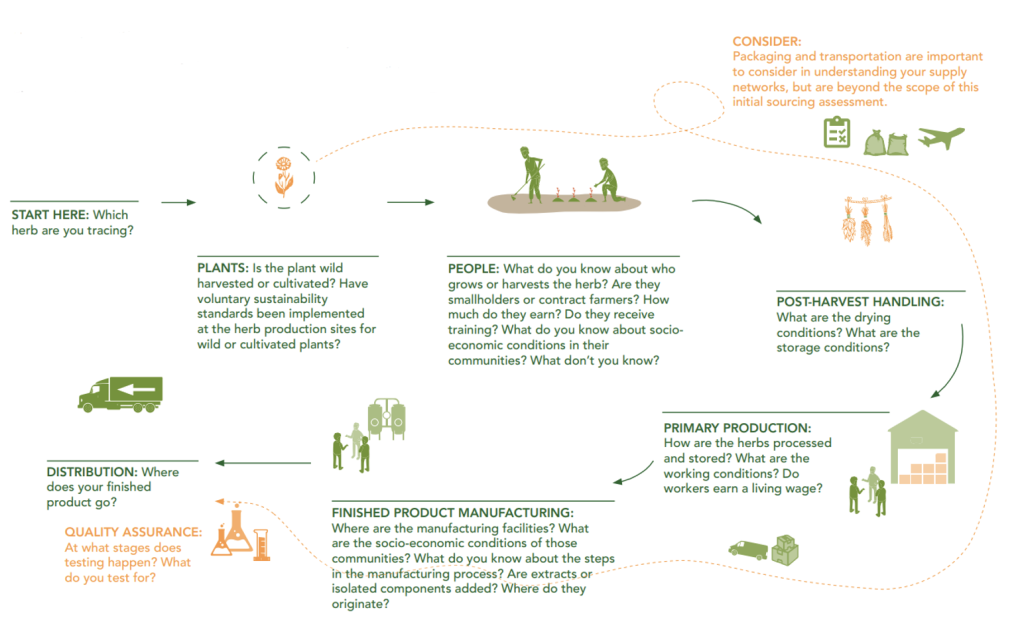
Source: Sustainable Herbs Program, 2023
Tips:
- Before joining any certification schemes, verify with potential buyers if your target market requires or demands certification and whether it will give you a competitive advantage over other suppliers. Examples of standards include EU organic certification and Fairtrade certification, as well as Fair for Life and FairWild.
- See the CBI study on buyer requirements for natural health products to Europe, as it provides information about the mandatory ethical sourcing and biodiversity requirements you must meet to enter the European market.
- See the CBI study entitled The EU Green Deal – How will it affect my business? for more information on the EU Green Deal and its implications.
- Make your supply chain transparent and your ingredients traceable. Use appropriate tracking and tracing or other audit and certification systems to show where your ingredients come from.
- Think about registering on platforms like Sedex to streamline your ethical sourcing documentation and show your commitment to responsible practices.
10. Increased need for traceability and transparency within supply chains
Traceability has become more important in the natural ingredients for health products industry. There is an increase in health-conscious and environmentally sustainable consumption. Because of this, consumers are starting to demand products with clear information regarding origin, ingredients and manufacturing processes. Simply explained, traceability involves the ability to trace the journey of a product from its source to its end-user. It ensures accountability and clear steps at each stage. Doing this allows a company to tell their customers the story behind its branded botanical ingredient.
This trend is becoming more important as a result of EU regulation on environmental sustainability. In particular, the European Green Deal and the Directive on corporate sustainability due diligence require European companies to identify, and where necessary prevent or mitigate, any adverse impacts along their supply chains in regard to human rights and the environment. This regulation means that importers will be obligated to carry out due diligence investigations on their suppliers. As an exporter, you will be required to provide information on your environmental and social impact.
This can be done by working on a traceability system, which offers several advantages. Trust-building is one of the most important aspects of international trade. Through traceability you can build credibility and loyalty. This leads to positive word of mouth recommendations. Transparent supply chains can make exporters stand out and attract environmentally conscious importers and consumers. Besides this, a traceability system can help you to identify potential issues related to contamination of your natural ingredients. Being able to identify issues will ensure product safety and compliance with rigid quality standards.
The use of artificial intelligence (AI) to improve supply chain traceability is on the rise and is expected to grow further. AI has the potential to enhance traceability systems in various ways. For example, data lies at the heart of traceability, and AI-powered data collection can improve data quality, encourage supplier participation and boost operational efficiency. AI can analyse vast volumes of data, identify patterns and make predictions. Within supply chains, it can be applied to demand forecasting, optimise logistics and detect anomalies in data that could indicate fraud or inefficiencies.
Tips:
- Learn how to set up a traceability system in natural ingredients supply chains by reading this article by the Initiative for Sustainable Agricultural Supply Chains.
- Remember that establishing a traceability system does not have to be too complicated. Exporters can design simple systems with their suppliers that document where the products are grown, who is involved, and their impact on the environment and society. To make the process simpler, you should assign codes to each producer and provide information on volume in each batch. The documentation process can be done on paper, but many businesses are implementing digital solutions to make traceability activities efficient.
- Certifications for your ingredients can help you prove the traceability of your products, as this is verified and documented by an independent third-party inspector. However, before obtaining certification and making investments, always check with your (potential) buyer if there is demand and interest in the certified ingredient.
ProFound – Advisers In Development carried out this study on behalf of CBI.
Please review our market information disclaimer.
Search
Enter search terms to find market research
